Are you searching for a comprehensive guide to ICP-MS (Inductively Coupled Plasma Mass Spectrometry)? This is the ultimate guide for you! We will cover everything from sample preparation to data analysis so you can get the most out of your ICP-MS analysis.
This blog will discuss the fundamentals of ICP-MS, how to prepare samples for analysis, and how to interpret the results. Furthermore, this will provide helpful tips and tricks to ensure you get accurate and reliable results every time. So if you’re ready to learn about ICP-MS, let’s get started!
Sample Preparation
Sample preparation is essential to the ICP-MS process and should not be noticed. It is vital to ensure the sample being analyzed is free from any interferences that could affect the accuracy of the results. There are several ways, including acid digestion, microwave digestion, solid phase extraction, and graphite furnace atomization.
Organic materials and acidic digestion are often used as samples. This involves adding hydrochloric or nitric acid to the sample to break the organic material into its elemental components. Microwave digestion is another option, which uses high-power microwaves to heat the sample and break it down into constituent elements.
Solid phase extraction is a sample preparation method that passes the sample through a series of columns containing adsorbent materials such as alumina, silica, and polystyrene. The adsorbent material collects the sample material, allowing it to be eluted in a solvent for further analysis.
Finally, graphite furnace atomization is a technique used for sample preparation that involves passing a sample through an intense heating element to vaporize it. This method allows for higher resolution and accuracy when analyzing samples with trace elements.
No matter what method of sample preparation is used, it is essential to ensure that the sample is free from any interferences that could affect the accuracy of the results. Proper sample preparation is vital for successful ICP-MS results.
Inlet System
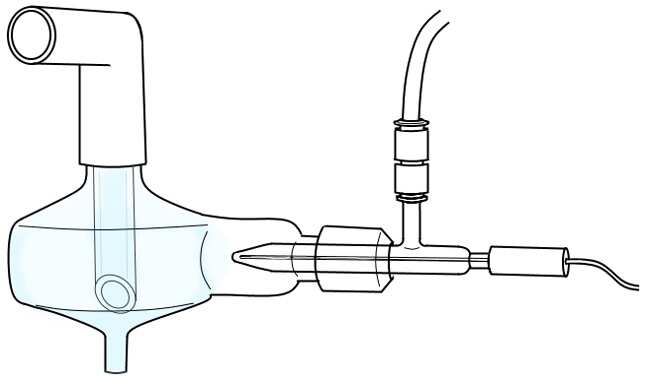
The inlet system is a critical component of an Inductively Coupled Plasma Mass Spectrometer (ICP-MS). The inlet system functions to introduce the sample solution into the plasma source. It must be designed to meet the sample introduction requirements of the specific ICP-MS application. Generally, the inlet system consists of a pump, nebulizer, desolvation, and a spray chamber.
The pump is usually a peristaltic pump which delivers a consistent flow rate. It supplies the sample solution to the nebulizer, which converts the sample solution into a fine aerosol mist. The aerosol is then directed into desolvation, vaporizing the droplets and reducing the vapor pressure.
The vaporized sample solution is then directed into the plasma source through the spray chamber. The force of the sample solution within the spray chamber should be regulated at 0.2-0.5 PSI for optimal performance.
The inlet system must be adequately maintained to ensure accurate results and minimize contamination. This includes regularly cleaning and replacing parts such as the nebulizer and desolvation to prevent clogging or breakage. Additionally, it is essential to avoid introducing impurities into the inlet system from any external sources.
Plasma Source
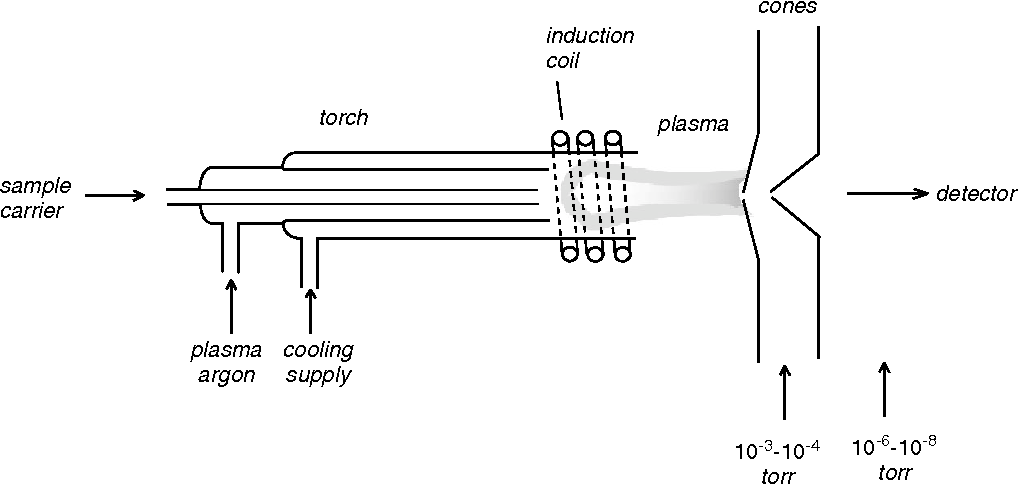
The plasma source is essential to ICP-MS, as it ionizes the sample for mass spectrometry. The plasma source is an electrically-driven device that produces an intense radio frequency (RF) electric field that causes the sample to become ionized. The plasma source is typically a torch or a channel.
The most common type of plasma source used in ICP-MS is the argon plasma torch, which uses argon gas to create a high-temperature, low-pressure plasma. The RF voltage applied to the argon gas causes electrons to move around rapidly in the gas, resulting in ionization. The argon plasma torch can be operated at temperatures between 5,000 and 8,000 K, producing a range of ions with different energies.
Another type of plasma source used in ICP-MS is the channel plasma source. A channel plasma source uses two electrically charged electrodes to create plasma within a tube or channel. The sample is introduced into the channel and then ionized by the plasma. Channel plasma sources typically operate at temperatures between 500 and 3,000 K and are more energy efficient than an argon plasma torch.
Proper maintenance includes regular cleaning and replacing components, ensuring the gas flow rates are correctly set, and adjusting the RF voltage. Additionally, it is essential to ensure that the sample is properly prepared before entering the plasma source, as any contaminants or molecules that enter the plasma source can cause contamination and affect the data quality.
Mass Spectrometer
A Mass Spectrometer is an analytical instrument used to identify a sample’s chemical composition by separating it into its parts and measuring the mass-to-charge ratio of each. The most commonly used type of Mass Spectrometer in ICP-MS is a Quadrupole Mass Spectrometer. This type of spectrometer uses four parallel rods to focus ions on an exit slit, which allows for a higher degree of sensitivity and specificity than other types.
Using a high voltage, the ions can be separated by their m/z values and detected by a detector, resulting in a signal that can be used to measure concentrations of elements or molecules. In addition to the Quadrupole Mass Spectrometer, other types of Mass Spectrometers, such as time-of-flight (TOF) and ion trap, are also used in ICP-MS.
Data Analysis
Data analysis is organizing, interpreting, and drawing conclusions from data collected during ICP-MS measurements. It is essential to accurately analyze the results to get meaningful conclusions and make significant decisions. Data analysis involves techniques such as graphical displays, hypothesis testing, statistical modeling, machine learning, and predictive modeling.
These techniques allow researchers to identify patterns in the data and make sound decisions based on those patterns. Additionally, data analysis helps confirm or reject hypotheses that may be used to inform future experiments.
To effectively analyze data collected from an ICP-MS instrument, it is essential to understand the data collection parameters, calibration methods, and the various data types that can be managed. Knowing how to interpret the data and draw meaningful conclusions from the results is also necessary.
Conclusion
In conclusion, the fundamentals of ICP-MS are powerful and efficient tools for analyzing various types of samples. It is capable of detecting trace elements in solutions, as well as providing elemental analysis of solid samples. The key to successful ICP-MS analyses is careful attention to sample preparation and data analysis techniques. With proper training, the possibilities are endless for what can be achieved with this fantastic technology. With its many applications and increasing popularity, it will remain a staple tool in laboratories for many years.








